Our experience and innovation provides the correct solutions
CONTRACT DEVELOPMENT ORGANIZATION
MabPlex offers a series of integrated CDO for mAb products from antibody humanization, cell line development, to analytic development, quality standard establishment, and stability studies.
Cell Line Development
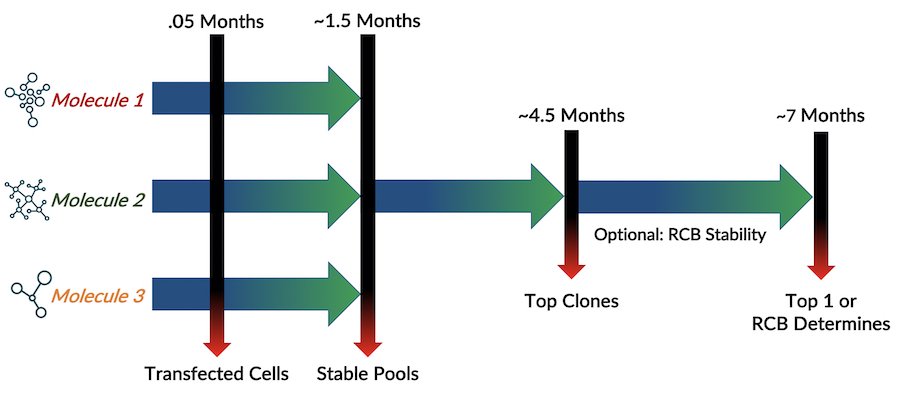
The establishment of a stable cell line is essential for large scale antibody drug production. MabPlex has experience with a broad range of mammalian expression platforms. CHO cell is primarily used for cell line development and a proprietary line has been established. These cell line platforms offer high titers and efficiency to meet challenging timelines. Relying on extensive experience and advanced equipment, MabPlex provides high-quality service in accordance with your specific needs.
- Experience in expression systems including our own proprietary expression platform
- High transfection efficiency
- High-throughput screening of clone selection
- Cell line stability evaluation
UPSTREAM PROCESS DEVELOPMENT
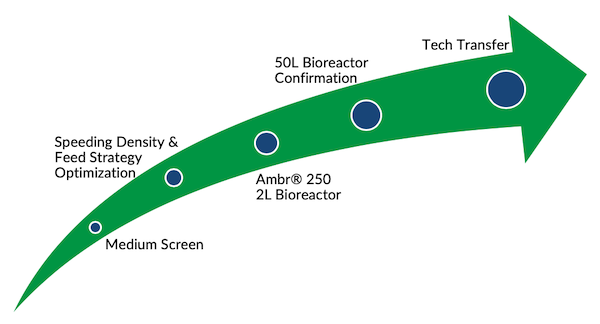
MabPlex provides upstream process service with various cell culture systems and advanced in-process analytical technologies, as well as extensive experience on various CHO cell types.
- Screening of CHO culture media
- Development and optimization of feeding strategy
- Clarification development (depth filtration, cetrifugation, TFF)
- Tech transfer and scale up of bioreactor process up to 2000L
- Material production
- Process characterization including by using Ambr® system or bench-top bioreactors
DOWNSTREAM PROCESS DEVELOPMENT
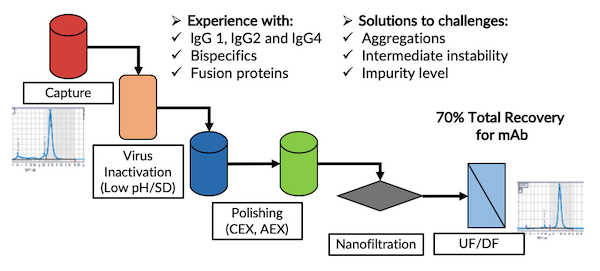
MabPlex offers downstream process services for a variety of biologics including monoclonal antibodies, recombinant proteins. The downstream process secures removal of process and product related impurities, high recovery.
- High throughput screening of chromatography resins
- Chromatography purification parameters optimization (Affinity, IEX, HIC and Mixed Mode)
- Viral clearance development
- Process characterization with scale-down model for chromatographic purification process and viral clearance studies
- Chromatography resin lifetime studies
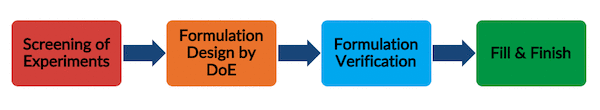
FORMULATION PROCESS DEVELOPMENT
- Efficient formulation development and optimization
- Long-term and accelerated stability
- Freeze thaw stability
- Agitation & shaking stability
- Lyophilization development, optimization and scale up
- Forced degradation including photo stability
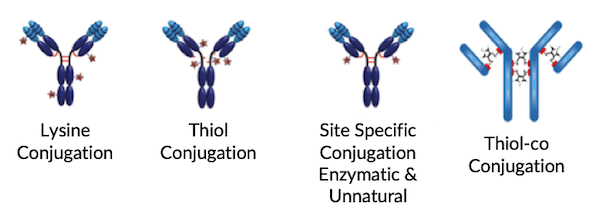
ANTIBODY DRUG CONJUGATION PROCESS DEVELOPMENT
MabPlex is the one of the leading companies for ADC drug development in the world which provides technically challenging service from development to commercial production including both payload and ADC. Additionally, innovative linker technologies are implemented to improve therapeutic window and decrease nonspecific cytotoxicity. The first-class service can significantly speed up ADC drug development process.
- Linker/toxin synthesis process and optimization
- ADC process development
- Intermediate and final API stability
- Analysis of impurities
- Analytical method development
- Multiple formats of ADCs
- Process characterization
- Successful scale-up experiences from milligram to kilogram scale
ANALYTICAL METHOD DEVELOPMENT
MabPlex applies state-of-the-art instrument to perform comprehensive characterization on biologics and linker/payload Analytical method development and optimization/improvement and validation of monoclonal antibody, recombinant protein, complex glycoprotein and ADC.
- Physicochemical characterization including but not limited to primary structure, higher order structure, PTMs, heterogeneity analysis etc.
- Bioactivity assay development, optimization, validation: Antigen-Binding activity, Fc-Receptor Binding activity, Cytotoxicity, Cell Growth Inhibition activity etc.
- ADC special characterization analysis: DAR, Conjugate site, Free drug
Small molecule project research: structure characterization, purity analysis, bioactivity analysis, etc.